 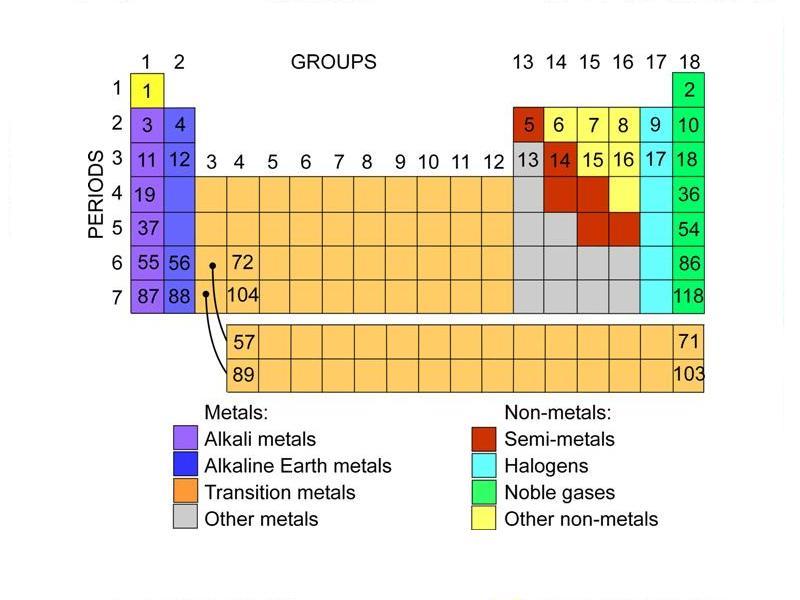 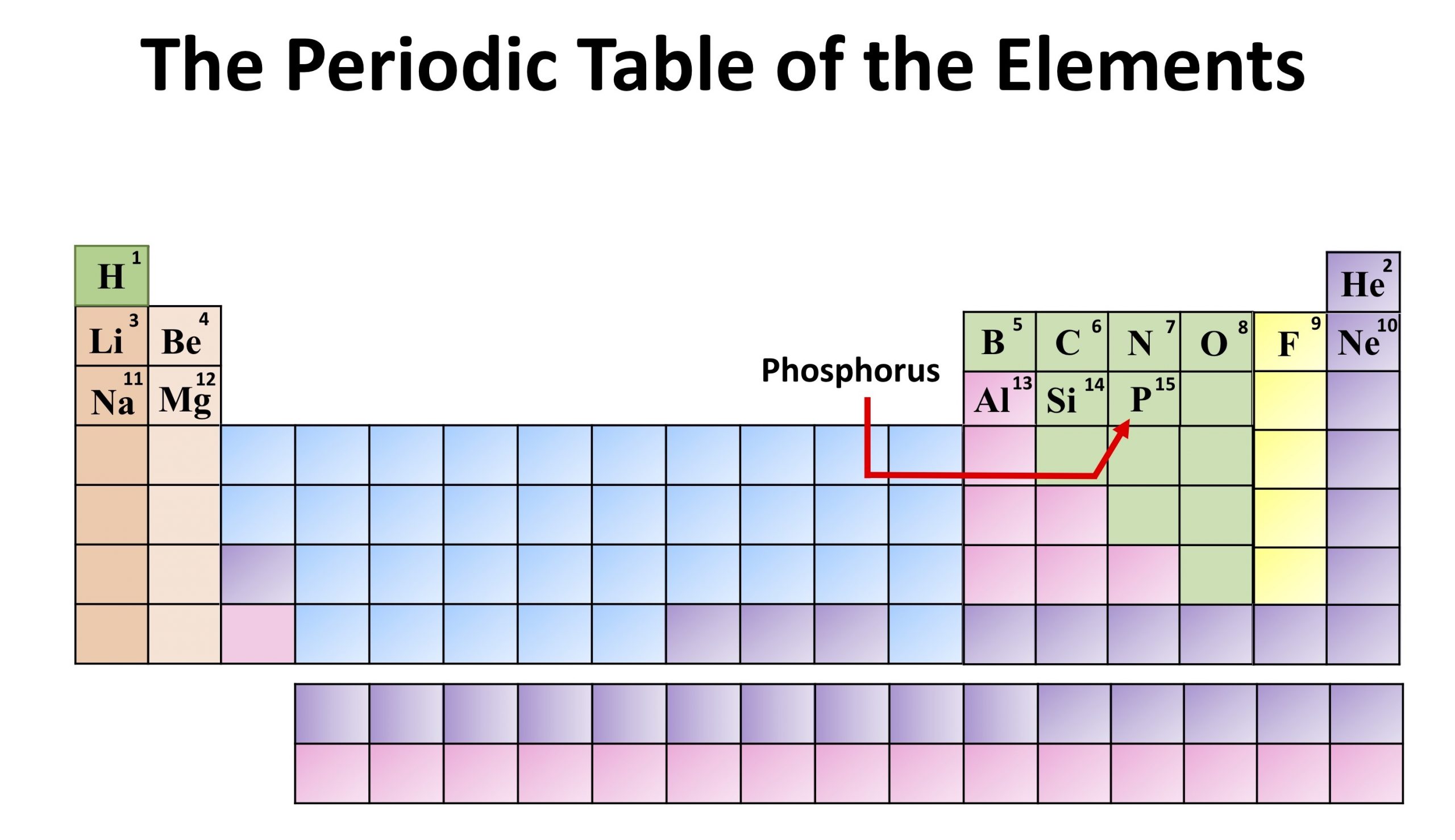
Element Lead (Pb), Group 14, Atomic Number 82, p-block, Mass 207.2. The elements in a group have very similar chemical properties, which arise from the number of valence electrons presentthat is, the number of electrons in the outermost shell of an atom. Periodic Table of Videos Created by video journalist Brady Haran working with chemists at The University of Nottingham. 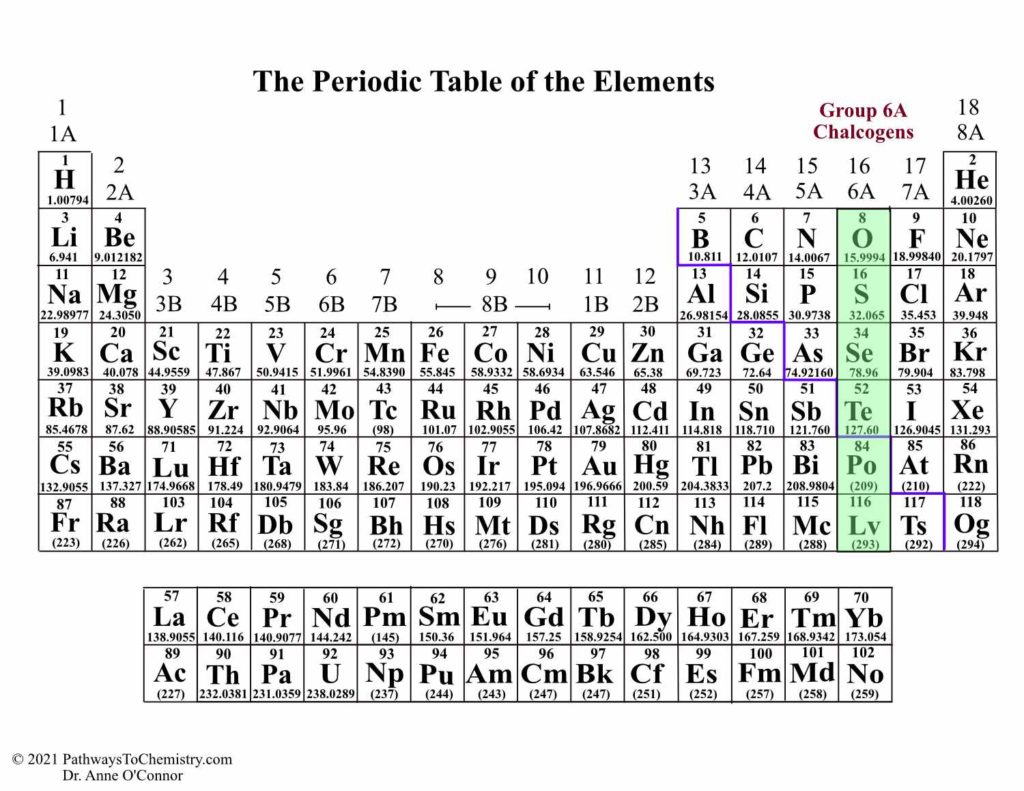
Not to miss out, element 102, Nobelium, No, is named in honor of Alfred Nobel, who set aside his vast fortune to establish Nobel Prizes. The groups of the periodic table are displayed as vertical columns numbered from 1 to 18. Mendeleev never received a Nobel Prize for his work, but element 101 was named Mendelevium, Md, after him. 
Wolfram Tungsten which is element 74 on the periodic table uses the symbol W for wolfram. William Zeise was a Danish chemist who synthesized the first organometallic compound. For instance they discovered phosphorus when they isolated it from urine. William A name of English origin and means with gilded helmet. Over time these gaps have gradually been filled in as scientists unearthed new elements. Not only did Mendeleev arrange the elements in the correct way, but he also had the foresight to leave gaps for undiscovered elements. At that time, he had only 50 elements to arrange. So I know I presented a lot of information to you right now, but just keep in mind we have our periods and we. So these are the different names for the groups of the periodic table. Play a video: Periodic Table: Group Names. He wrote the properties of the elements on pieces of card and rearranged them until he realised that, by putting them in order of increasing atomic weight, certain properties of elements regularly occurred. Atoms and the Periodic Table Periodic Table: Group Names. Then in 1869, a Russian scientist called Dmitri Mendeleev produced one of the first practical periodic tables. This resource contains complete, up to date, colour coded (and black and white) Periodic Table, Mendeleev, Newland, Solid, Liquid and Gas, Metals and Non-metals, Groups (Actinides/Lanthanide etc), Electron Configuration (for the first 20) and s, p, d, f orbitals and Mass Number/Atomic Number. Atoms in a period have the same number of electron shells. Name of the eight groups in the periodic table: Alkali metals Alkaline earth metals Rare. Periods are the horizontal rows that run from left to right, while groups are the vertical columns (going from up to down). Several other attempts were made to group elements together over the coming decades. Science, Tech, Math Science Periodic Table of Element Groups GIPhotoStock / Getty Images By Anne Marie Helmenstine, Ph.D. An element period is a horizontal row on the periodic table. Periodic Table: The periodic table is a system for arranging elements on the basis of their physical and chemical properties. The earliest attempt to classify the elements was in 1789, when Antoine Lavoisier grouped the elements based on their properties into gases, non-metals, metals and earths. The discovery of other elements followed regularly and soon it became necessary to arrange them in some sort of order. However, the first scientific discovery of an element occurred in 1649 when Hennig Brand discovered phosphorous. \): The periodic table organizes elements with similar properties into groups.Copper has been used by humans for as much as 7000 years and elements such as gold, silver, tin, lead and mercury have been known for many thousands of years. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
